Bio-Optronics is the creator of Clinical Conductor (CC CTMS), known for generating significant savings with its robust finance capabilities, it is also the leading CTMS for executing collaboratively across multiple clinical trials and sites, including enabling remote workflows. Used by more research sites around the world than any other CTMS, Clinical Conductor is currently managing over 40,000 studies, including a significant percentage of the world’s COVID-19 clinical trials.
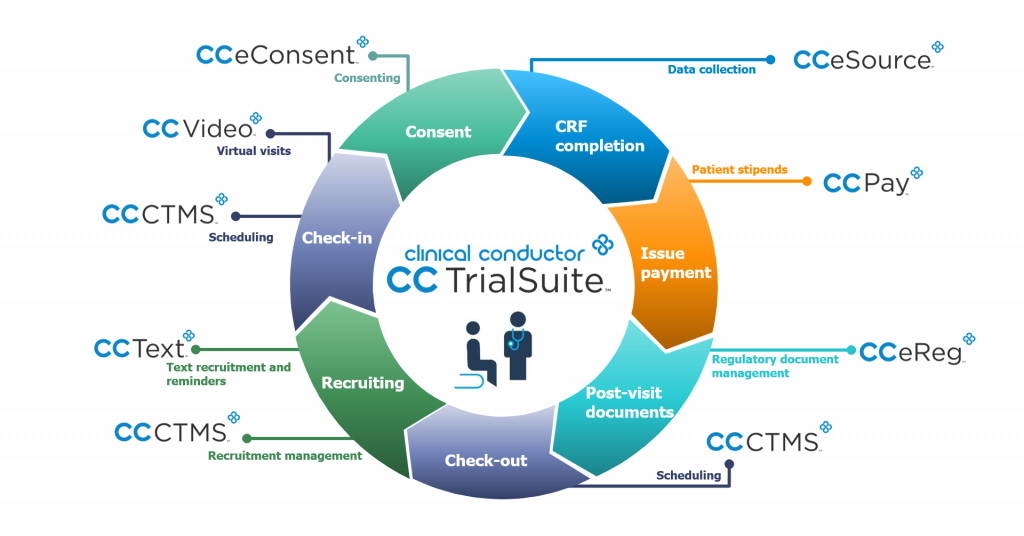
CC CTMS is part of the CCTrialSuite, a comprehensive set of integrated, patient-centric clinical research management products that together streamline research operations while maintaining a high degree of compliance. Complementing Clinical Conductor in the suite are several remote workflow enabling solutions. CCPay offers sites the unprecedented ability to both control finances and improve patient satisfaction with real-time payment reimbursements using secure pre-paid payment cards, all controlled remotely from CC CTMS dashboards. While CCText, the only fully CTMS-imbedded, conversational 2-way text messaging solution, has demonstrated clear results with increased patient recruitment, engagement and retention. An expanding set of remote workflow capable solutions include Part 11 compliant CCeReg document management, remote monitoring enabled CCeSource data capture, mobile friendly CCeConsent, and easy to engage remote visits using the HIPAA compliant CCVideo solutions from a single SaaS supplier, Bio-Optronics.
Click here for free access to The Ultimate Guide to Clinical Trial Software!
Also visit www.bio-optronics.com or follow @BioOptronics on Twitter or LinkedIn
Products
CC CTMS
CC CTMS is the only clinical trial management system with strong financial and compliance capabilities that is designed to accommodate the unique needs of your organization across multiple sites and countries. Run the world’s best research.